Water - It's everywhere!
Click here to go over the basics of this essential molecule of life
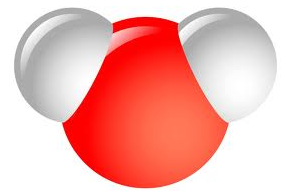
Water is Polar - So What's with the Mickey Mouse?
Water is formed when 2 hydrogen and 1 oxygen are bonded together. The hydrogen (the ears) have a slightly positive charge and the oxygen has a slightly negative charge. These polar ends cause a bonding that is strong and allows water to have unique solvent qualities.
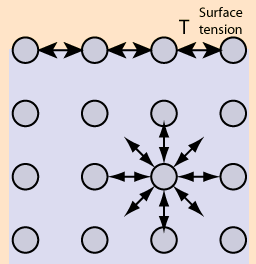
Surface Tension -
Cohesion and Surface Tension are the cohesive forces between molecules in a liquid that are shared with all neighboring atoms. Those on the surface have no neighboring atoms above, and exhibit stronger attractive forces upon their nearest neighbors on the surface. This enhancement of the intermolecular attractive forces at the surface is called surface tension.
SO... Why does the belly flop hurt so much?!
Simple - surface tension. :)
Simple - surface tension. :)
Meniscus
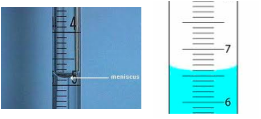
When measuring water in a scientific lab or even in the kitchen you will encounter a meniscus.
The meniscus is the curve of the liquid in the container that is caused by adhesion and cohesion.
The graduated cylinder to the immediate left is read as 66 since that is where the bottom of the curve is in the graduated cylinder.
This property of water causes us to read our measurements very carefully to make sure we are correct!
Why do the cohesive properties of water matter to life?
Ever seen a tree take a walk to the water fountain?!
Cohesion is how plants "drink." In a plant, there are straw like structures that allow for movement of nutrients and water to flow through the plant called xylem and phloem. The cohesive nature of water allows for these liquids to move inside the plant.
Ever seen a tree take a walk to the water fountain?!
Cohesion is how plants "drink." In a plant, there are straw like structures that allow for movement of nutrients and water to flow through the plant called xylem and phloem. The cohesive nature of water allows for these liquids to move inside the plant.
What is a solution, a solute and solubility?
Sometimes a picture is worth 1000 words - click below for a clear description...
Mixtures - What type did you mean?

In science there are all types of mixtures - here are two we often talk about...
Heterogeneous and Homogeneous.
Heterogeneous and Homogeneous.
Still Confused? Love Water? Find more info here...
What you need to know:
You need to know the specific properties of water that contribute to Earth's suitability as an environment for life. These include:
- cohesive behavior
- ability to moderate temperature
- expansion upon freezing
- versatility as a solvent
- hydrogen bonding and polarity
Example One
Water is essential for life. Its special properties make water the single most important molecule in plant life. Which of the following properties of water enables it to move from the roots to the leaves of plants?
A. Water expands as it freezes.
B. Water is an excellent solvent.
C. Water exhibits cohesive behavior.
D. Water is able to moderate temperature.
Example Two
Large bodies of water, such as lakes and oceans, do not quickly fluctuate in temperature. What is the reason for this phenomenon?
A. Water is an acid.
B. Water is a versatile solvent.
C. Water has a high heat capacity.
D. Water acts as a buffer.
Example Three
Why does ice stay at the top of oceans instead of sinking to the bottom?
A. Ice is colder than liquid water.
B. Ice is less dense than liquid water.
C. Ice is more dense than liquid water.
D. Ice is warmer than liquid water.
Example Four
Water is often called the "universal solvent" because many substances can be dissolved in water. What property of water allows it to be such a versatile solvent?
A. purity
B. polarity and cohesion
C. high heat capacity
D. expansion upon freezing
Water is essential for life. Its special properties make water the single most important molecule in plant life. Which of the following properties of water enables it to move from the roots to the leaves of plants?
A. Water expands as it freezes.
B. Water is an excellent solvent.
C. Water exhibits cohesive behavior.
D. Water is able to moderate temperature.
Example Two
Large bodies of water, such as lakes and oceans, do not quickly fluctuate in temperature. What is the reason for this phenomenon?
A. Water is an acid.
B. Water is a versatile solvent.
C. Water has a high heat capacity.
D. Water acts as a buffer.
Example Three
Why does ice stay at the top of oceans instead of sinking to the bottom?
A. Ice is colder than liquid water.
B. Ice is less dense than liquid water.
C. Ice is more dense than liquid water.
D. Ice is warmer than liquid water.
Example Four
Water is often called the "universal solvent" because many substances can be dissolved in water. What property of water allows it to be such a versatile solvent?
A. purity
B. polarity and cohesion
C. high heat capacity
D. expansion upon freezing